How to convert pressure gauge reading to temperature?
Mastering water temperature in an espresso machine boiler is essential for achieving perfect extraction. Thanks to the Clapeyron formula, it is possible to convert the pressure measured by a gauge into water temperature, and thus optimize your machine's performance.
This article offers you a clear explanation of the principle, a precise conversion table, as well as a practical method for applying these calculations on a daily basis.
As I briefly explained in my previous article, it is possible to determine water temperature by measuring the boiler pressure.
Basic principle:
- Water boils at 100 °C at an atmospheric pressure of 1.013 bar (sea level).
- The higher the altitude, the lower the atmospheric pressure, and therefore the boiling temperature decreases (e.g. at 6000 m: 80 °C at 0.471 bar).
The theory
In the case of a boiler (e.g. espresso machine), the water temperature can be determined by knowing the altitude and internal pressure.
This relationship is calculated using the Clapeyron formula:
$$ dp = \frac{L}{T (\Delta V)} \times \frac{dT}{T} $$
Legend:
- T: Temperature
- ∆V: Variation in molar volume during phase change
- L: Latent heat (enthalpy variation at constant pressure)
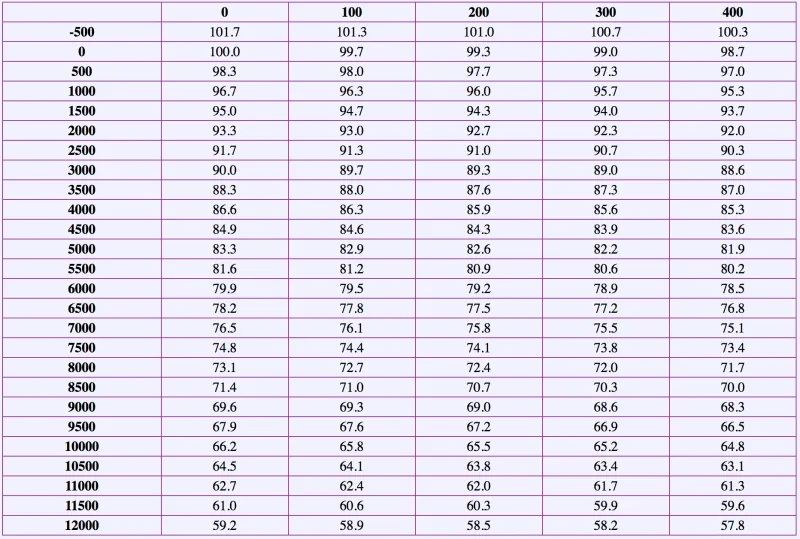
Using this formula, I created a pressure/temperature conversion table for an altitude of 0 m (sea level).
Barista tip: ask your roaster for the ideal extraction temperature for your coffee. Some coffees are better at 88 °C, others at 92 °C.
Water Boiling Temperature According to Altitude
Practice: simple method
For everyday use, a free Android app, Nomograph, automatically calculates your boiler temperature.
Instructions:
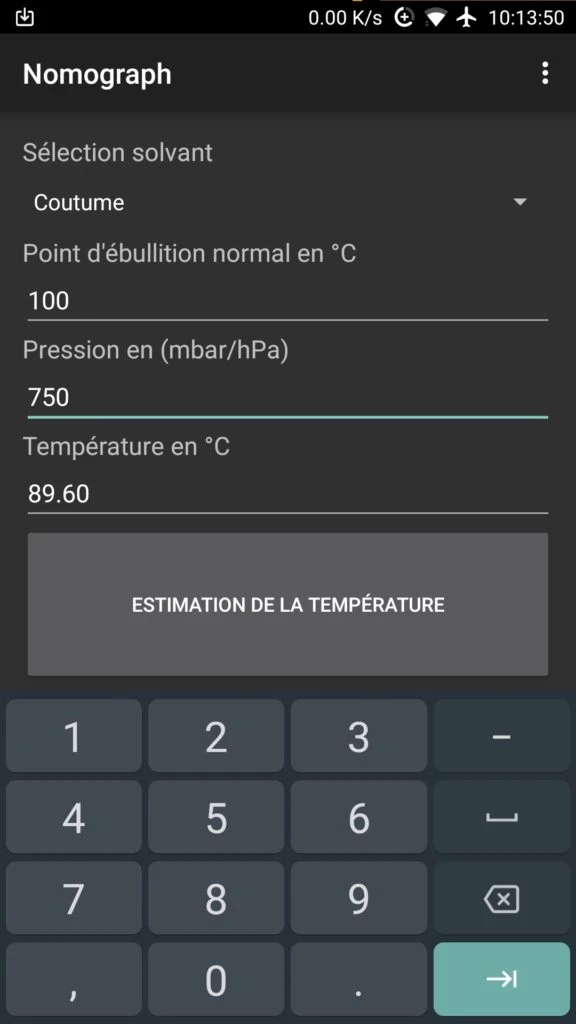
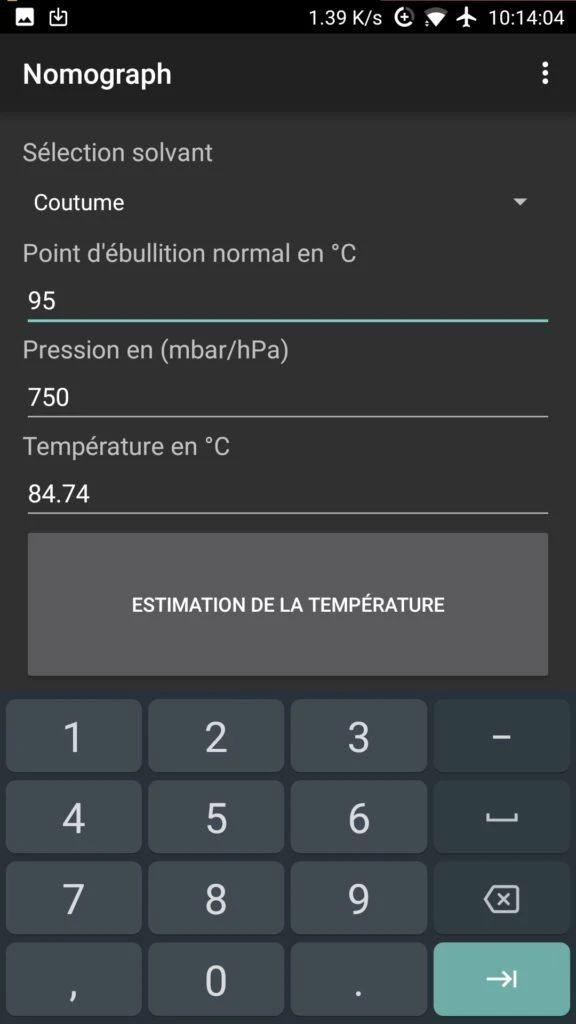
- In "Normal Boiling Point in °C", enter your water's boiling temperature according to altitude (100°C at 0 m, 95°C at 1500 m).
- In "Pressure in mbar/hPa", enter the pressure reading from the gauge (e.g. 0.750 bar → 750 mbar).
- Click on "Temperature Estimation" to display the actual temperature.
Calculation Example
You can also do the reverse: enter a desired temperature to get the corresponding pressure.
For precise control, note your reference temperatures on a sheet next to your machine. This will allow you to quickly adjust your pressure for each espresso.